AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Different blocks in periodic table8/17/2023  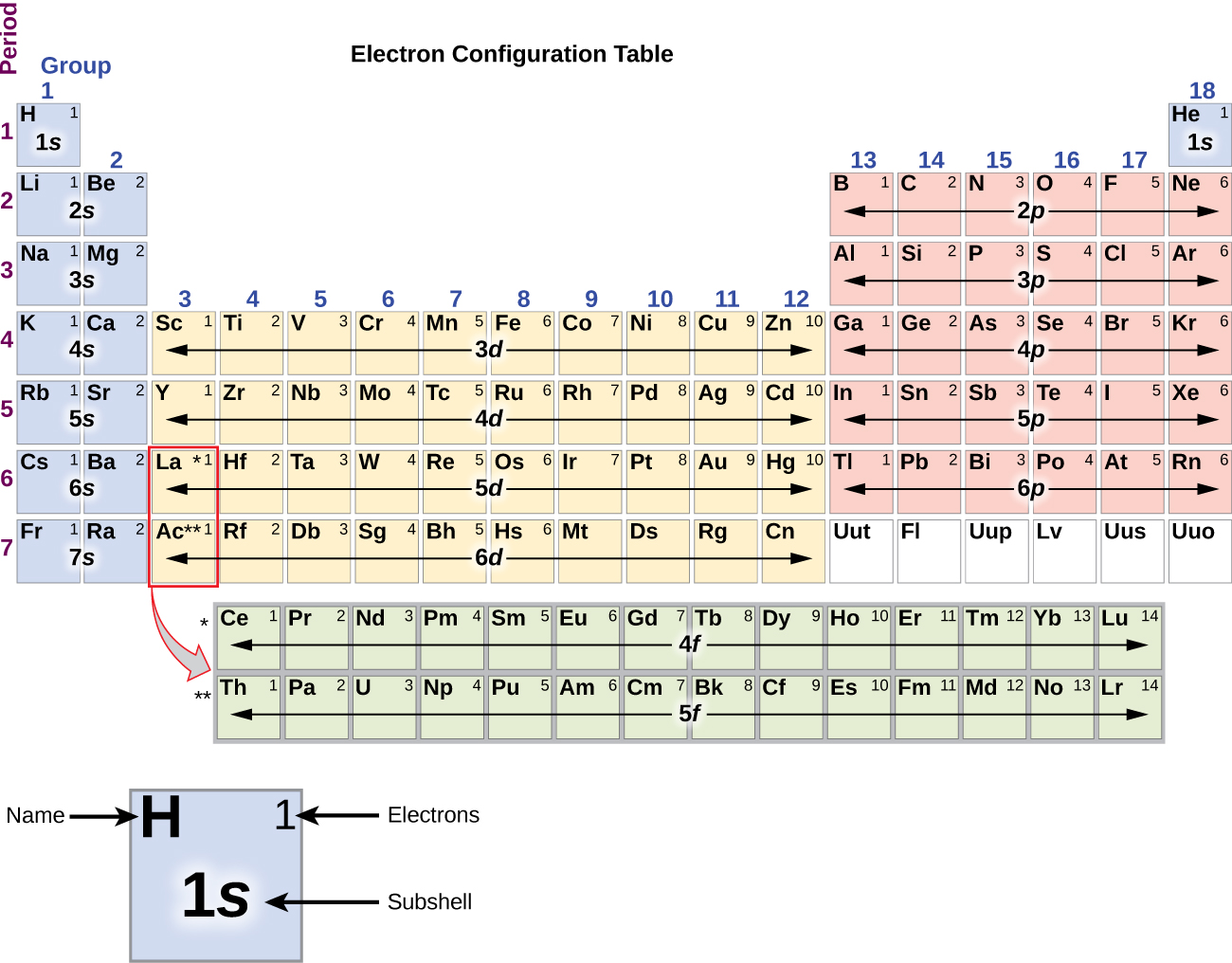 
The number of isotopes What happens when the electron moves from the first energy level to the second energy level? Energy is absorbed, and an emission line is produced.Įnergy is released, and an emission line is produced. The number of protons and neutrons in an atom The difference between the number of protons and electrons The sum of electrons and protons in an atom What is the formula of a compound formed between nitrogen (N) and barium (Ba)? What is a newly industrialized nation? 1) a country with an agrarian based economy, but social and political progressĢ)a country with a more advanced economy, but not the same social and political progressĤ)a country with social and political progress, but lacking an advanced economic system What information does the atomic mass of an element provide? A. The components of a heterogeneous mixture are spaced evenly throughout. The components of a homogeneous mixture are evenly distributed. The number of ingredients in each will differ. Help What is the main difference between a homogeneous mixture and a heterogeneous mixture? A. What is the percentage of nitrogen in N2O? Show all calculations leading to an answer. Keywords: Property, intensive, extensive, physical properties, chemical properties, density, substance, amount, quantity, nature, molarity, units, m, b, mol/L, mol/volume, molar mass. The main purpose of conducting experiments: /question/5096428 Rate of chemical reaction: /question/1569924Ģ. So its unit can be calculated as follows:ġ. The unit of b is equal to that of m(density). Substitute 58.5 g/mol for the molar mass of NaCl in equation (4). So two quantities on the right-hand side of equation (2) must have the same units and equation (2) becomes, Quantities with same units are added, subtracted, multiplied or divided. Substitute the formula of given quantities in equation (1). The formula to calculate the molarity of the solution is as follows: It is denoted by M and its unit is mol/L. Molarity is a concentration term that is defined as the number of moles of solute dissolved in one litre of the solution. The formula to calculate the density of a substance is, Both mass and volume are the physical properties that are extensive in nature and their ratio comes out to be an intensive quantity that depends only on the nature of the substance, not on the amount of the substance. Mass, enthalpy, volume, energy, size, weight, and length are some of the examples of extensive properties.ĭensity is defined as the ratio between mass and volume. These are additive in nature when a single system is divided into many subsystems. These are the properties that depend on the amount of the substance. Temperature, refractive index, molarity, concentration, pressure, and density are some of the examples of intensive properties. Their values remain unaltered even if the system is further divided into a number of subsystems. These don't depend on the size of the system. 
These are the properties that depend on the nature of the substance. The property is a unique feature of the substance that differentiates it from the other substances. 
M₂/ V₂ is the density of water: 1.00 g/mLĬomparing with the equation Molarity = m×density b, you obtain: Making the simplistic assumption that the dissolved NaCl(s) does not affect the volume of the solvent water means 1000Vt = V₂ Molarity = number of moles / Volume of solution in litersĦ) Substitute in the equation of density: ⇒ mass in grams = number of moles × molar mass Number of moles = mass in grams / molar mass \) (Credit: Christopher Auyeung and Joy Sheng Source: CK-12 Foundation License: CC BY-NC 3.2) Density of the solution: mass in grams / volume in mililiters 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
